 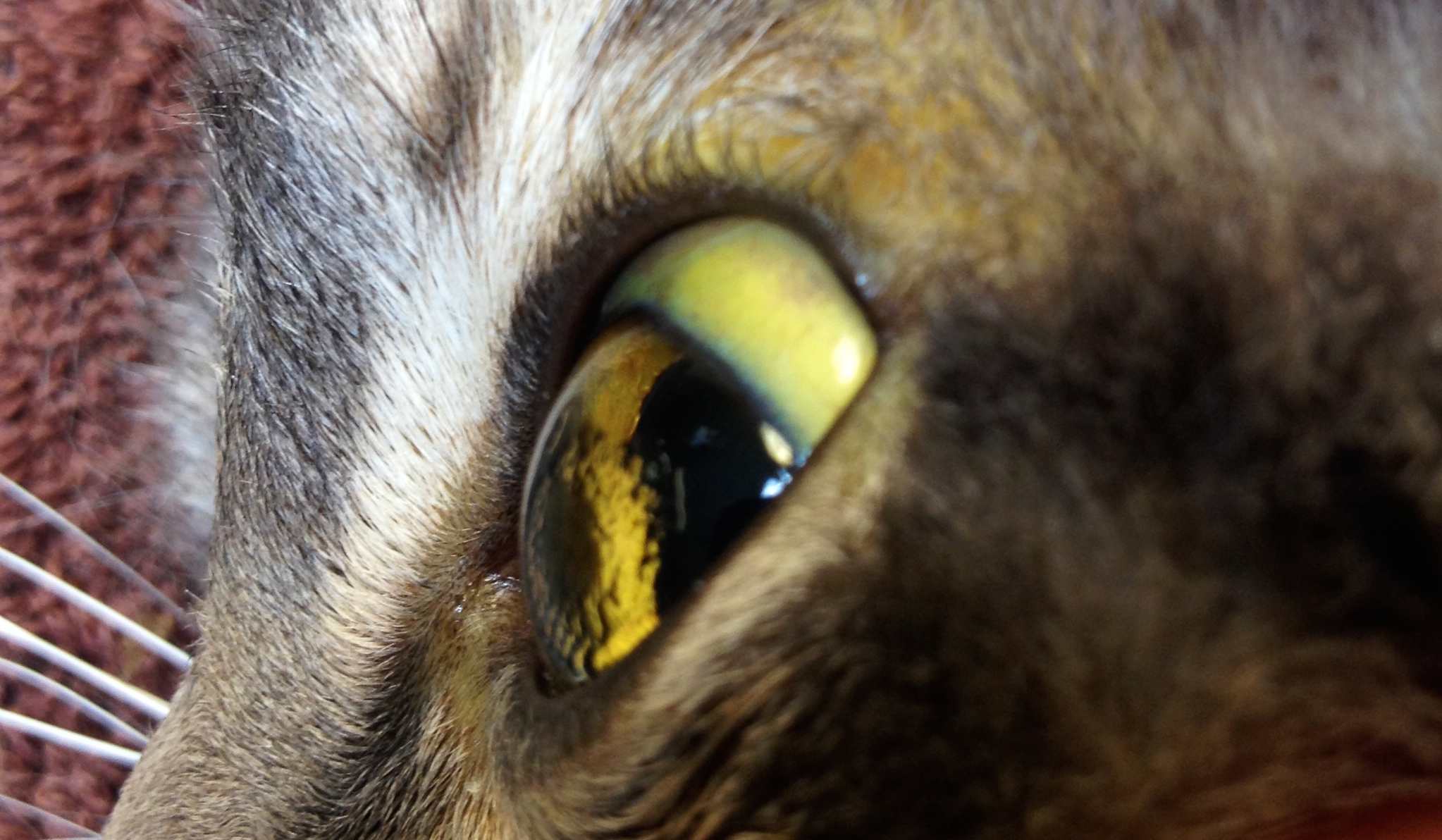 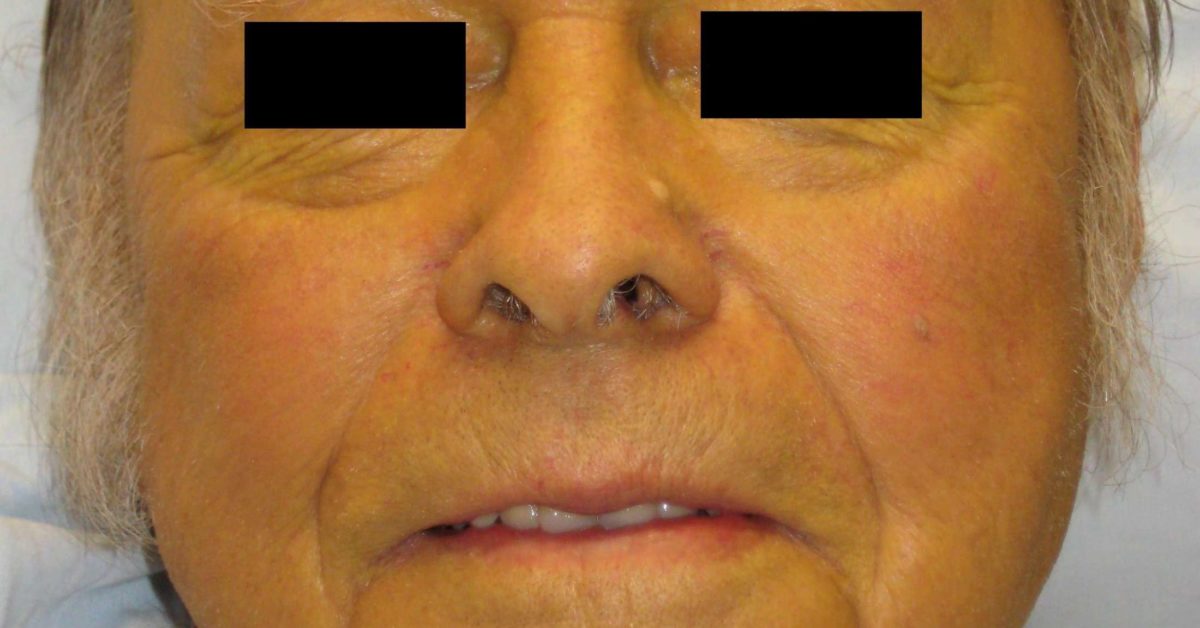
Please see full Prescribing Information for PYRUKYND. Hepatic Impairment : Avoid use of PYRUKYND in patients with moderate and severe hepatic impairment. P-gp Substrates: Avoid concomitant use with substrates that have narrow therapeutic index.UGT1A1 Substrates: Avoid concomitant use with substrates that have narrow therapeutic index.Carrier proteins transport bilirubin into the liver, where it is conjugated and excreted in the bile. Sensitive CYP3A, CYP2B6, CYP2C Substrates Including Hormonal Contraceptives: Avoid concomitant use with substrates that have narrow therapeutic index. This occurs when: bilirubin > 2 mg/dl (Normal 0.00.3mg/dl) Feline Bilirubin Metabolism o Bilirubin is a heme breakdown product, produced mostly in the spleen.If there are no alternatives, adjust PYRUKYND dosage. The first step that expert doctors take is to observe the human eye and understand the severity. Moderate CYP3A Inducers: Consider alternatives that are not moderate inducers. Scleral Icterus Disease: Treatment and Identifying Method.Moderate CYP3A Inhibitors: Do not titrate PYRUKYND beyond 20 mg twice daily. 
Two thirds of the patients treated by irradiation alone had no problems with vision or difficulties related to treatment of. Strong CYP3A Inhibitors and Inducers: Avoid concomitant use. Unilateral scleral icterus is a very rare finding.Plasma elevation of both unconjugated and conjugated bilirubin due to. Abstract Jaundice is a condition in which the skin, whites of the eyes and mucous membranes turn yellow because of a high level of bilirubin, a yellow-orange bile pigment. Classification and causes of jaundice or asymptomatic hyperbilirubinemia. Varices are monitored with endoscopy and often require prophylaxis with nonselective beta blockers. It can be observed on scleral examination. Treatment goals are preventing cirrhosis, decompensation, and death. In the ACTIVATE trial, the most common adverse reactions including laboratory abnormalities (≥10%) in patients with PK deficiency were estrone decreased (males), increased urate, back pain, estradiol decreased (males), and arthralgia. Scleral icterus is due to bilirubin deposition in the conjunctiva and subsequently in scleral stroma. When discontinuing treatment, monitor patients for signs of acute hemolysis and anemia including jaundice, scleral icterus, dark urine, dizziness, confusion, fatigue, or shortness of breath.Īdverse Reactions : Serious adverse reactions occurred in 10% of patients receiving PYRUKYND in the ACTIVATE trial, including atrial fibrillation, gastroenteritis, rib fracture, and musculoskeletal pain, each of which occurred in 1 patient. Gradually taper the dose of PYRUKYND to discontinue treatment if possible. PYRUKYND is a pyruvate kinase activator indicated for the treatment of hemolytic anemia in adults with pyruvate kinase (PK) deficiency.Īcute Hemolysis : Acute hemolysis with subsequent anemia has been observed following abrupt interruption or discontinuation of PYRUKYND in a dose-ranging study. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
